Precise and contamination-free injections
The Puringe project is developing a novel syringe system for precise and contamination-free injections, with an initial focus on intravitreal drug delivery (IVOD). The core innovation is a flexible, impermeable membrane that physically separates the active ingredient from the syringe, thereby reliably preventing contamination from silicone oil or abrasive particles.
Intravitreal drug administration using the Puringe, which enables precise and particle-free delivery.
© Copyright: Charité - Universitätsmedizin Berlin
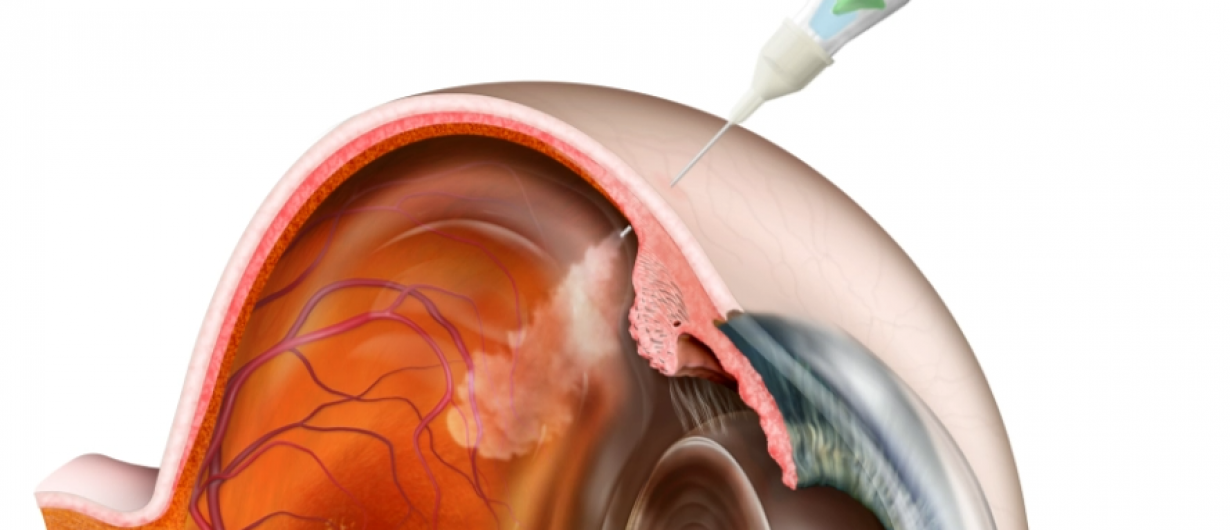
At the same time, the Puringe functions as an integrated dosing aid and is designed to prevent overdoses and unnecessary medication waste. The solution is compatible with existing clinical routines, can be combined with standard syringes and needles via a Luer-Lock connection, and is suitable for mass production.
Thanks to the use of cyclo-olefin copolymer (COC), the Puringe is also suitable for sensitive active ingredients and pre-filled applications, including storage at very low temperatures. In addition to IVOM, the Puringe thus also addresses other long-term application areas such as mRNA, gene, and cell therapies.
The goal of the project is to ensure regulatory compliance through further development up to clinical application and to prepare for rapid market translation for the benefit of patients and the healthcare system.
The project is funded by the Else Kröner-Fresenius Foundation.
