Image-based personalised prediction of residual risk and prognosis for cardiovascular and cerebrovascular diseases
Über das Projekt
The aim of the project is to investigate and quantify blood damage caused by turbulent mechanisms. A new experimental model – a turbulence-generating Taylor-Couette system (TCS) comprising two counter-rotating cylinders – will be used to study blood damage experimentally. In this TCS, the inner and outer cylinders can rotate independently of one another. When both cylinders rotate in opposite directions at a specific combination of rotational speeds, a uniform turbulent flow is generated in the gap. In particular, by selecting appropriate rotational speeds for both cylinders, it is possible to generate a laminar flow with the same mean shear rate as that prevailing in the turbulent regions within the same apparatus.
This allows the blood damage caused by turbulent effects to be compared with the blood damage caused by the widely studied laminar flow. The flow is experimentally investigated using a two-component LDA measurement in a blood substitute fluid. The characteristic turbulence parameters are determined as a function of rotational speed, and the influence of flow state, turbulence intensity and exposure time on these parameters is examined.
The variation in the measured parameters will also be investigated. To this end, blood samples from animals and subsequently from healthy volunteers will first be introduced into the TCS gap and analysed using standard methods. A correlation between blood damage and the turbulent flow parameters will then be analysed.
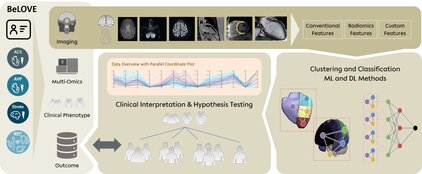
Cardiovascular disease (CVD) is the leading cause of death worldwide. Risk factors such as diabetes, high blood pressure and high cholesterol are used to predict the risk of cardiovascular disease and to plan prevention strategies. People who have already suffered a heart attack, stroke or heart failure are particularly at risk. In these patients, the risk of recurrent events varies greatly, which is why a more accurate assessment of risk is highly desirable.
New biomarkers could help to better identify individual risks and optimise treatment. In patients with cardiovascular disease, there is a wide range of comorbidities, which complicates the prognosis. Furthermore, diseases affecting other organs, such as the kidneys or brain, influence the course of heart disease. These interrelationships have not yet been sufficiently researched, but they are important for developing better treatments. Current research is therefore focusing on personalised approaches that, based on technologies such as ‘omics’ and artificial intelligence, make individual risks and disease mechanisms more measurable.
The project ‘Image-based personalised prediction of residual risk and prognosis for cardiovascular and cerebrovascular diseases based on mapping data from the BELOVE study’ is funded under SPP 2177: Radiomics: The Next Generation of Medical Imaging. Professors Dr Anja Hennemuth, Univ. Prof. Dr Jeanette Schulz-Menger and Dr Kersten Villringer jointly lead and supervise the research project.
Projektinformationen
Keywords
Kennzahlen
DFG
Ihr Ansprechpartner
Anja Hennemuth, Prof. Dr.-Ing.